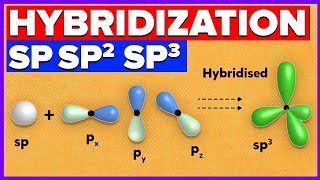
Web Reference: Hybridization was introduced to explain molecular structure when the valence bond theory failed to correctly predict them. It is experimentally observed that bond angles in organic compounds are close to 109 o, 120 o, or 180 o. In chemistry, orbital hybridisation (or hybridization) is the concept of mixing atomic orbitals to form new hybrid orbitals (with different energies, shapes, etc., than the component atomic orbitals) suitable for the pairing of electrons to form chemical bonds in valence bond theory. Simply put, hybridization is the way that distinct atomic orbitals combine together to form identical hybrid orbitals which can participate in bonding much more favorably than unhybridized ones.
YouTube Excerpt: This organic chemistry video tutorial explains the
Net Worth Profile Overview
Hybridization Sp2 Orbitals Net Worth 2026: Salary, Income & Wealth Net Worth & Biography
Estimated Worth: $4M - $22M
Salary & Income Sources

Career Highlights & Achievements

Assets, Properties & Investments
This section covers known assets, real estate holdings, luxury vehicles, and investment portfolios. Data is compiled from public records, financial disclosures, and verified media reports.
Last Updated: April 3, 2026
Net Worth Outlook & Future Earnings

Disclaimer: Disclaimer: Net Worth estimates are based on publicly available data, media reports, and financial analysis. Actual numbers may vary.